
DREYER Erwin
- UMR Silva (Inrae, UL, AgroParisTech), INRAE, Champenoux, France
- Functional forest ecology , Tree biology and physiology
- administrator, manager, recommender
Recommendations: 7
Reviews: 0
Recommendations: 7
The diversity of radial variations of wood properties in European beech reveals the plastic nature of juvenile wood
Recommendation of an interesting analysis of ontogenic and adaptive variations in local wood properties in European Beech (Fagus sylvatica L.)
Recommended by Erwin Dreyer based on reviews by Régis Fichot and 1 anonymous reviewerThe mechanics of wood as a material for construction, furniture, pulp and other uses have been addressed in a very large number of papers and are a well-established field for both research and technical applications (for example among many others, see Pöhler et al., 2006 for beechwood). In addition to such approaches that derive from material sciences, further developments based on similar physical concepts addressed the questions raised by the biomechanics of the standing and the growing tree which requires some degree of postural control and sensing of specific signals (gravity, movements…; see for instance Fournier et al. 2013; Dlouha et al., 2025). Within this field of research, the question of the correlation of wood anatomy (diameter of xylem tracheids or vessels, fibre content and angles, vessel wall thickness… ) and biomechanical properties is of prime importance, and specific responses of wood and bark components have been identified over the last decades. In particular the occurrence of reaction wood generates local strains and contributes to the postural control (Ruelle, 2014).
In this preprint, Almeras et al. address a complementary question related to the properties of juvenile wood in trees. During the first years of the growth of young trees, the annual tree rings display quite specific properties (large tree rings, less dense wood, …) that gradually change with age and dimensions of the trees until reaching a range of values typical for adult trees. During the first years, the interannual changes might follow an ontogenetic trajectory mainly related to age (and dimensions) while in later stages, they appear to be strongly controlled by environment (wind, soil fertility, site index, irradiance, water availability, ...). All these changes result in radial profiles along tree rings (from the pith to the bark) of three main features that govern the biomechanical properties of wood, namely the width of the annual tree ring, the local specific gravity (wood density), and the specific modulus which contributes with density to the local modulus of elasticity (Fournier et al. 2013). Such gradients of local wood properties within stems have been analysed and synthesised in the last years (Lachenbruch et al. 2011, Meinzer et al. 2014).
Here, the authors address the question of local variations of such properties within tree stems as a function of the distance to the pith (inversely related to the age of the trees when the ring was formed) in a broadleaved species, European Beech (Fagus sylvatica L.). They checked whether ring width, specific gravity and specific modulus display systematic trends from pith to bark across tree stems, and whether these trends enable the detection of a general ontogenic (age-related) effect with very similar patterns in juvenile wood of different individuals, or whether adaptive factors (modulated by the environment and by the mechanical constraints induced by the postural control of growth) dominate already in juvenile wood, like it does at later stages. Such questions were already analysed in the wood of some coniferous species (softwood with tracheids), but less frequently in hardwood species (angiosperms, like Beech with its diffuse porous wood anatomy).
Before starting the analysis of age-related tree ring properties in juvenile wood, the authors addressed the potential impact of duraminisation, which affects the oldest tree rings in the inner wood (that is those formed during the juvenile growth stages). Duraminisation results from local deposition of a number of secondary metabolites and results in the build-up of heartwood; in the case of beech however, reddish heartwood is less present than in other species (Knoke, 2003). Almeras et al showed here that the occurrence of reddish wood did only marginally affect the mechanical properties and contributed only marginally to the observed variations among trees
The very solid experimental design enabled the authors to clearly assign a fraction of the observed variation in the three parameters to (i) the site where trees had grown, (ii) to the individuals within these sites and (iii) to the position of the ring within the stem. The intraindividual component of the variation was much larger than the former. However, the observed asymmetry in the patterns of ring properties in juvenile wood, and the large variability in these patterns among trees led the authors conclude that the ontogenic juvenility effects, visible in ring width were largely dominated by other effects influenced by the local environment. In this respect, the results differ from those that were recorded earlier with Pinus taeda L. in a plantation (i.e., trees of the same age and homogenous spatial distribution, Bendtsen and Senft, 1986).
The recommended version of the preprint is very original as it shows how local (radial) variations of biomechanical wood properties can be addressed in a systematic way. This lead to novel approaches that share light on the processes governing wood formation in trees.
The first version of the preprint was submitted over a year ago. The recommended version differs in many respects from the initial one. The two rounds of reviews with external reviewers, and the additional one with the recommender resulted in an in-depth reorganisation of the statistical analysis and of the demonstration. This took some time, but shows also the benefits that may be gained during an open peer review process like the one developed by the Peer Community in….
References
Bendtsen BA, Senft J. 1986. Mechanical and anatomical properties in individual growth rings of plantation-grown eastern cottonwood and loblolly pine. Wood Fiber Sci 18: 23-38.
Dlouhá J, Moulia B, Fournier, M et al. 2025 Beyond the perception of wind only as a meteorological hazard: importance of mechanobiology for biomass allocation, forest ecology and management. Ann For Sci 82, 1. https://doi.org/10.1186/s13595-024-01271-6
Fournier M, Dlouha J, Jaouen G, Alméras T. 2013. Integrative biomechanics for tree ecology: beyond wood density and strength. J Exp Bot, 64, 4793-4815. https://doi.org/10.1093/jxb/ert279
Knoke T. 2003 Predicting red heartwood formation in beech trees (Fagus sylvatica L.)? Ecol. Model. 169, 289-312. https://doi.org/10.1016/S0304-3800(03)00276-X
Lachenbruch, B., Moore, J.R., Evans, R. (2011). Radial Variation in wood structure and function in woodypPlants, and hypotheses for its occurrence. In: Meinzer, F., Lachenbruch, B., Dawson, T. (eds) Size- and age-related changes in tree structure and function. Tree Physiology, vol 4. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-1242-3_5
Longuetaud F, Mothe F, Santenoise P, Diop N, Dlouha J, Fournier M, Deleuze C. 2017. Patterns of within-stem variations in wood specific gravity and water content for five temperate tree species. Ann For Sci 74:64. https://doi.org/10.1007/s13595-017-0657-7
Meinzer, F., Lachenbruch, B., Dawson, T. (eds). 2014. Size- and age-related changes in tree structure and function. Tree Physiology, vol 4. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-1242-3_5
Pöhler E, Klingner R, Künniger T. 2006. Beech (Fagus sylvatica L.) - Technological properties, adhesion behaviour and colour stability with and without coatings of the red heartwood. Ann For Sci 63: 129-137. https://doi.org/10.1051/forest:2005105
Ruelle J. 2014. Morphology, anatomy and ultrastructure of reaction wood. In: Gardiner B, Barnett J, Saranpää P, Gril J (eds) The Biology of Reaction Wood. Springer Series in Wood Science. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-10814-3_2
Cite the recommended preprint:
Almeras Tancrède, Jullien Delphine, Liu Shengquan, Loup Caroline, Gril Joseph, Thibaut Bernard (2025) The diversity of radial variations of wood properties in European beech reveals the plastic nature of juvenile wood. HAL, ver.6 peer-reviewed and recommended by PCI Forest and Wood Sciences https://hal.science/hal-04133248
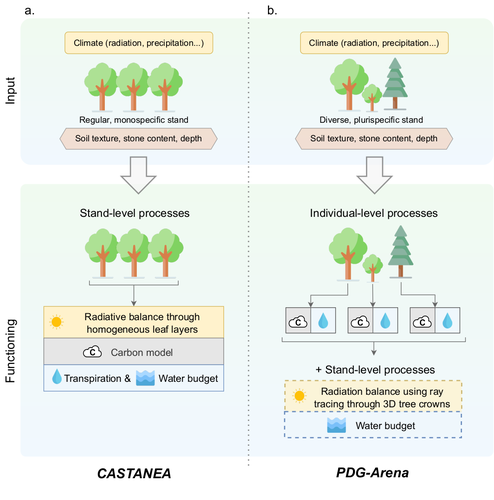
PDG-Arena: An ecophysiological model for characterizing tree-tree interactions in heterogeneous and mixed stands
Is it possible to model and predict the growth of mixed-forests under a changing climate : some answers provided by the new model « PDG-Arena » developed on the CAPSIS platform.
Recommended by Erwin Dreyer based on reviews by Harald Bugmann and 1 anonymous reviewerThis interesting preprint develops a new model with a quite strange name (see below why this name was used by the authors) that aims at describing the growth of mixed forests (it may also be used for monospecific forests with a regular or irregular structure). It is based on two main features: it is a process-based model that integrates a tree-tree interaction module. The model derives from the modelling framework « Physio-demo-genetics model PDG » developed earlier at INRAE-Avignon, and « Castanea », developed at Orsay and Avignon as a widely used stand-level process-based model. « Arena » underlines the competition between trees in mixed stands. The « PDG-Arena » model was developed on the Capsis platform (https://capsis.cirad.fr/capsis/presentation), which is a shared platform for the development of tree growth models under a very open framework maintained by several French institutions.
The reviewers and the recommender saw two important strengths in the preprint : (i) PDG-Arena is definitely a significant improvement when compared to the existing models and may be used for accurately predicting the growth and dynamics of mixed stands ; (ii) it was validated against an impressive data set gained in a quite impressive network of mixed stands of beech (Fagus sylvatica L.) and silver fir (Abies alba Mill.) with a 5-year growth analysis at individual tree level (based on two detailed surveys at individual tree level. PDG-Arena could properly predict the growth of these stands and showed significant improvements for the existing models.
During the review process, many important questions were raised, some directly related to the manuscript (which the authors addressed very convincingly during the revision of the first version of the preprint), and some refering to more general debates around predictive forest growth models able to integrate long-term changes. For instance, the long-lasting debate opposing the proponents of process-based forest growth models (that might require a large number of sometimes difficult-to-document processes leading to growth predictions, Forrester et al. 2016) and phenomenological models that directly link actual (recorded) growth and stand dynamics to climate and soil variables, is not yet settled. Both approaches display strengths and weaknesses. Similarly, the ability of both families of models to predict the impact of extreme events induced by climate change (like severe drought episodes associated to high temperatures likely to happen at higher frequencies in the near future) remains to be assessed and is the main challenge for this area of research.
A second question that raises large interest is whether mixed forest stands display a larger resilience to future climate changes than monospecific and homogenous stands. Camille Rouet and his colleagues present convincing results that this is the case (both using their model and from the recorded data). However, there is still debate whether the observed over-yielding in mixed forests under favourable conditions will be maintained under severe drought episodes and after a return to more favourable water availability and moderate temperatures (Jourdan et al, 2019; Jourdan et al, 2020).
The model is available for testing under the condition of participating to the CAPSIS community (which gives access to the shared resources available on the platform. The codes and data used to test the performance of the model can be accessed on the Zenodo data repository.
This preprint should open the way for fruitful cooperation and further improvements in a very important area for forest science in the near future.
References
Camille Rouet, Hendrik Davi, Arsène Druel, Bruno Fady, Xavier Morin (2024) PDG-Arena: An ecophysiological model for characterizing tree-tree interactions in heterogeneous and mixed stands. bioRxiv, ver.3 peer-reviewed and recommended by PCI Forest and Wood Sciences https://doi.org/10.1101/2024.02.09.579667
Jourdan, M., Kunstler, G., Morin, X., 2020. How neighbourhood interactions control the temporal stability and resilience to drought of trees in mountain forests. Journal of Ecology 108, 666–677. https://doi.org/10.1111/1365-2745.13294
Jourdan, M., Lebourgeois, F., Morin, X., 2019. The effect of tree diversity on the resistance and recovery of forest stands in the French Alps may depend on species differences in hydraulic features. Forest Ecology and Management 450, 117486. https://doi:10.1016/j.foreco.2019.117486
Forrester, D.I., Bauhus, J., 2016. A Review of Processes Behind Diversity Productivity Relationships in Forests. Current Forestry Reports 2, 45–61. https://doi:10.1007/s40725-016-0031-2
Mey, R., Zell, J., Thürig, E. et al. , 2022. Tree species admixture increases ecosystem service provision in simulated spruce- and beech-dominated stands. European Journal of Forest Research 141, 801–820 (2022). https://doi.org/10.1007/s10342-022-01474-4
Demographic and genetic impacts of powdery mildew in a young oak cohort
An interesting analysis of the interactions between English oaks (Quercus robur L.) and powdery mildew (Erisyphe sp.) at tree population level.
Recommended by Erwin Dreyer based on reviews by Louis Bernier and 1 anonymous reviewerThis preprint is recommended after a round of reviews by two external reviewers and the recommender and a careful revision of the initial text. The revision addressed all the the concerns raised by the reviewers and myself acting as recommender. I commend the authors for the care taken in the revision process and in addressing all concerns raised by the referees. The preprint is now in a status anabling a very positive recommendation, and I am convinced that PCI friendly journals will be keen to publish it given the quality of the contribution to Forest Pathology and Epidemiology as well as to and Forest Ecology.
Indeed, the preprint adresses an important topic in forest ecology and forest management. Powdery mildew (due to a complex of fungi species of Erysiphe spp.) is a very frequent pathogen affecting oaks and mainly English oak (Quercus robur L.), a widespread species in Western European forests which bears great ecological and economic interests (see Marçais and Desprez-Loustau, 2014, for a review). Young regenerations are mostly affected by the disease, which infects young, unfoldling leaves and leads to severe reductions in photosynthesis (Hajji et al, 2009) and in some cases to tree dieback in young regenerations, and sometimes in older trees in the case of repeated infestations over several years, or combinations with defoliaitions by processionnary moths (Thaumetopoea processionea L.). Powdery mildew was introduced to Europe at the beginning of the 20th century and caused initially very severe damage in oak stands; currently, damage is much less prevalent, probably due to a co-evolution of the oak host and the pathogen (Desprez-Loustau and Marçais, 2019).
The present investigation adds very interesting and important information to our current knowledge of this disease, and addresses in particular the genetic variability of susceptibility to the disease among oak families and the effect of the disease on the survival of seedlings in the long run. Five main research questions were addressed: i. How does powdery mildew affect juvenile survival; ii. Is the survival rate differing among oak families? iii. Does powdery mildew infection reduce the differences of fitness among oak families? iv. Does powdery mildew impact the genetic diversity of oak populations? v. Are there significant genetic associations between some genetic loci and seedling survival? These questions are important for unerstanding the evolution of a pathosystem like the powdery mildew/English oak system and for explaining the past dynamics of this pathosystem, which resulted in a dicrease in virulence of the disease since its introduction in Europe.
The preprint reports results from a very original and solid experimental design based on the cultivation in the field of 15 oak progenies comprising 1733 indivduals over a quite long time span (9 years) and recurrent observations of growth, survival, infection intensity, ... A control group was protected against the pathogen by application of a fungicide. Moreover, a large number of individuals were genotyped, using single nucleatide polymorphism (SNP) allowing the detection of some candidate loci, and preparing future genome-wide association studies. The results are quite clear, and add very important elements to our understaning of this interesting and evolving pathosystem present in mots of the western European oak forests.
This preprint is of particular interest since such approaches, which are becoming common in cultivated crops, have only seldom been applied to natural pathosystems despite their importance for the dynamics of forest ecosystems under the increasing impact of climate change. In brief, this is an important preprint that brings a large set of new data and addresses the urgent question of epidemiology of diseases in forest ecosystems.
References
Barrès B, Dutech C, Saint-Jean G, Bodénès C, Burban C, Fiévet V, Lepoittevin C, Garnier-Géré P, Desprez-Loustau M-L (2024) Demographic and genetic impacts of powdery mildew in a young oak cohort. bioRxiv, 2023.06.22.546164, ver. 2 peer-reviewed and recommended by Peer Community in Forest and Wood Science. https://doi.org/10.1101/2023.06.22.546164
Desprez-Loustau ML, Hamelin FM, Marçais B (2019) The ecological and evolutiionary trajectory of oak powdery mildew in Europe. In: Wilson K, Fenton A, Tompkins D, Wildlife Disease Ecology: Linking Theory to Data and Application. Ecological Reviews. Cambridge University Press, 978-1-107-13656-4.
https://doi.org/10.1017/9781316479964.015
Hajji M, Dreyer E, Marçais B. (2009) Impact pf Erysiphe alphotoides on transpiration and photosynthesis in Quercus robur leaves. Eur J Plant Pathol, 125, 63-72,
https://doi.org/10.1007/s10658-009-9458-7
Marçais B, Desprez-Loustau ML (2014) European oak powdery mildew: impact on trees, effects of environmental factors, and potential effects of climate change. Ann For Sci, 71, 633-642,
https://doi.org/10.1007/s13595-012-0252-x

Modelling the growth stress in tree branches: eccentric growth vs. reaction wood
An important contribution to the description of growth stresses in branches of adult trees based on a new model and an optimisation process with digitised branches.
Recommended by Erwin Dreyer based on reviews by Jana Dlouha and 1 anonymous reviewerThis interesting article (van Rooij et al, 2023) proposes an innovative modelling approach to the question of the biomechanics of a growing branch. The main aim is to model the “growth stress” (Fournier et al, 2013) it is exposed to while developing its radial structure in response to increasing weight. The proposed model is very interesting and novel with respect to the existing literature on this important topic in tree biology. The model bases on two major components of the structure of a growing branch: the eccentricity (the branch is usually thicker vertically than horizontally, which may provide the strength to resist the weight) and the production of reaction wood (Barnett et al, 2014) on one side of the branch which produces asymmetric forces against gravity. The reaction wood is either tension wood (in hardwood trees, e.g., angiosperms) or compression wood (in softwood trees, e.g., gymnosperms). The model is clearly described and based on a number of explicit and already described concepts with some simplifications (no local irregularities like nodes or holes, only vertical bending taken into account, branch growing straight at a constant angle, …) whose potential effects are nicely discussed and on a reliable and detailed set of analytical equations. The model addresses the dynamic changes resulting from branch growth, i.e., mainly radial growth which results in an accumulation of wood and in increasing mass and “growth stress”.
The model is tested during a virtual experiment using a small set of data from a large pine tree (taken as an example of a softwood conifer tree) and a cherry tree (taken as an example of a hardwood tree). The optimisation test uses the mean allometric values from 30 branches of each individual tree as an entry to the model. This test of the optimality of the model is a very useful prerequisite for the adoption of the model. One might however argue that some replicate examples from other tree species would have been welcome to better represent the potential inter-specific variability in the two groups (softwoods vs. hardwoods). Indeed, there is a lack of suitable data available to properly test the underlying hypotheses under different conditions (growth angles, wood densities, growth rate, branch aging, ….). However, the presented computations allow testing the plausibility of the model and of its main conclusions, with respect to some “growth stress” values reported in the literature. The results confirm that the contribution of reaction wood is dominant, even if the eccentricity of the branches bears a significant contribution in the two tested cases.
The present preprint has the potential to act as the foundation for some additional research that might challenge its main conclusions and provide (hopefully) more support to the main conclusion that eccentricity plays a minor but still significant role in ensuring the stability of the growing branches and that the main stabilising effects are produced by reaction wood.
This version of the preprint is now suitable for a recommendation. However, it still suffers a few minor typos and language issues that the authors might correct during further steps in the publication process (a final version as a preprint, or submission to a journal chosen by the authors). Among those typos, the fact that Prunus avium is a cherry tree and not a birch. Similarly, several references need be corrected and completed, and more care should be in general given to the scientific species names….
In conclusion, this modelling exercise and the optimisation procedure used here underline once more the importance of reaction wood as a stabiliser of the three-dimensional architecture of trees not only in the trunk (where it has been studied in detail), but also in the lateral and sometimes quite heavy branches.
Anyway, I believe this preprint (and the version potentially published in a journal) will become an important reference for future research about the biomechanics of branches and of tree crowns in general, and that it will trigger further research in this direction.
REFERENCES
Arnoul van Rooij, Eric Badel, Jean-François Barczi, Yves Caraglio, Tancrede Almeras, and Joseph Gril. (2023) Modelling the growth stress in tree branches: eccentric growth vs. reaction wood. HAL, ver. 4 peer-reviewed and recommended by Peer Community in Forest and Wood Science. https://hal.science/hal-03748026v4
Mériem Fournier, Jana Dlouha, Gaëlle Jaouen, Tancrède Almeras (2013). Integrative biomechanics for tree ecology: beyond wood density and strength. Journal of Experimental Botany, 60 (15), pp.4397-
4410. https://doi.org/10.1093/jxb/ert279
J.R. Barnett, Joseph Gril, Pekka Saranpää (2014) Introduction, In: The Biology of Reaction Wood, Springer Series in Wood Science, Springer (pub), Gardiner B., Barnett J., Saranpää P., Gril J (eds), p. 1-11. https://doi.org/10.1007/978-3-642-10814-3_1

Variability in seeds’ physicochemical characteristics, germination and seedling growth within and between two French Populus nigra L. populations
Black poplar (Populus nigra L.) seed physiology: an important issue for the in situ conservation of this riparian species
Recommended by Erwin Dreyer based on reviews by Tomasz A. Pawlowski, Eduardo Notivol and 1 anonymous reviewerSeed physiology, which was a little forgotten in forest ecology since several decades, needs be revitalized as a research area given the many open questions about population dynamics and demography in rapidly changing environments (and not only for endangered species).
Indeed, seed physiology was long mobilized mainly to optimize seed conservation and germination for the production of plant material in a range of tree species used for afforestation/plantation. In the case of back poplar (which by the way is the male genitor of the multiple hybrid Populus x euramerica poplar cultivars), the focus is rather on the conservation ecology of this riparian species, and mainly on in situ conservation (Lefèvre et al, 1998). Indeed, the protection of populations of Populus nigra L requires an improved understanding of the ecology of this species with a focus on reproduction. Indeed, black poplar seeds need to be rapidly disseminated, to germinate as soon as the conditions are favourable (with rather small time windows) and establish seedlings with access to water in the rather harsh environment of mobile and sandy river banks submitted to alternating periods of flooding and of severe water deficits during low river flows in summer (Imbert and Lefèvre, 2003; Corenblit et al, 2014; Tinschert et al, 2020).
This process is therefore central to the propagation/maintenance of these populations that are threatened by the destruction of river banks and by introgression by either genes from the widespread “Italica” cultivar of black poplar, of from other poplar species leading to a variety of natural hybrids (Smulders et al, 2008).
Many questions remain open about seeds of black poplar (Michalak et al, 2015). One of the most intriguing one is to what extent seed properties and physiology differ within and among local populations from different river catchments. This question was addressed in this preprint by Lefebvre et al. (2021) that provides a very detailed and comparative analysis of two populations from central and southern France, each represented by 10 half sib families (i.e., seed collected separately from 10 adult individuals after open pollination).
Investigated properties were mainly seed biomass, anatomy, germination rate, root growth, lipid and sugar contents, protein content (with identification of some major protein families).
The within populations variability was indeed quite large, but nevertheless there were significant differences between the two populations in several traits, like seed weight, lipid content, and starch content. Storage proteins differed among families, but only slightly between the two populations. However, the main conclusion was that intrinsic qualities of the seeds were not critical for early stage establishment in the two populations, despite some significant differences in mean seed biomass, in lipid and in soluble sugars contents.
The preprint nicely analyses these differences, brings a large set of new observations about the seed physiology of Populus nigra. The referees found the data produced during this research quite important and original. This is why, despite the fact that the number of tested groups of populations remains rather small and the link with seedling establishment remains rather weak, this study is an important contribution to conservation ecology. This research (and that of many other groups) needs be further developed with an emphasis on inter and intra population variation and on demogenetics of forest tree species.
References
Corenblit D., Steiger J., González, E et al. (2014), The biogeomorphological life cycle of poplars during the fluvial biogeomorphological succession: a special focus on Populus nigra L.. Earth Surf. Process. Landforms, 39: 546-563. doi: https://doi.org/10.1002/esp.3515
Imbert E. and Lefèvre F. (2003) Dispersal and geneflow of Populus nigra (Salicaceae) along a dynamic river system. Journal of Ecology 91: 447-456. doi: https://doi.org/10.1046/j.1365-2745.2003.00772.x
Lefebvre M., Villar M., Boizot N., Delile A., Dimouro B., Lomelech A.-M. and Teyssier, C. (2021) Variability in seeds’ physicochemical characteristics, germination and seedling growth within and between two French Populus nigra populations. arXiv, 2008.05744, ver 3 peer-reviewed and recommended by Peer community in Forest and Wood Sciences. https://arxiv.org/abs/2008.05744
Lefèvre F., Légionnet A., de Vries S. and Turok J. (1998) Strategies for the conservation of a pioneer tree species, Populus nigra L., in Europe. Genetics, Selection, Evolution 30, S181-196. doi: https://doi.org/10.1186/1297-9686-30-S1-S181
Michalak M., Plitta B.P., Tylkowski T. et al. (2015) Desiccation tolerance and cryopreservation of seeds of black poplar (Populus nigra L.), a disappearing tree species in Europe. European Journal of Forest Research 134, 53–60. doi: https://doi.org/10.1007/s10342-014-0832-4
Smulder M.J.M., Beringen R., Volosyanchuk R. et al. (2008) Natural hybridisation between Populus nigra L. and P. x canadensis Moench. Hybrid offspring competes for niches along the Rhine river in the Netherlands. Tree Genetics & Genomes 4, 663–675. doi: https://doi.org/10.1007/s11295-008-0141-5
Tinschert E., Egger G., Wendelgass J. et al. (2020) Alternate reproductive strategies of Populus nigra influence diversity, structure and successional processes within riparian woodlands along the Allier River, France. Journal of Hydro-environment research 30, 100-108. doi: https://doi.org/10.1016/j.jher.2020.03.004

High-quality SNPs from genic regions highlight introgression patterns among European white oaks (Quercus petraea and Q. robur)
A new resource for a better understanding of the complex genetic dynamics within the oak syngameon
Recommended by Erwin Dreyer based on reviews by Oliver Brendel, Ricardo Alia, Komlan Avia and Hilke SchröderWhen we launched the multidisciplinary PCI Forest and Wood Sciences, we were well aware that this PCI devoted to a broad object (forests represent 1/3 of the emerged land area) and gathering multiple disciplines (from biology to social sciences), would be confronted with some challenges with respect to the range of pre-prints we would have to consider. Among the first ones submitted to the new PCI was this very interesting one (Lang et al, 2021) in the area of tree genomics and tree population genetics with a strong background in bioinformatics. This could well have been submitted to the PCI in Genomics, or the PCI in Evolutionary Biology, but finally fits very well in PCI Forest and Wood Sciences due to its finality: provide reliable genomic resources to tackle questions that are central to our understanding of the dynamics of forest ecosystems and the adaptation of trees to local conditions.
The phylogenetic status of the two sympatric oak species Quercus robur L. and Q. petraea Matt. Liebl. is an important question that was the center of in depth investigations by the BioGeCo research group at Bordeaux in cooperation with many European groups. The two species are major forest tree species in western Europe, structuring important ecosystems and providing high quality wood and other important ecosystem services. These species are considered to build up what is now called a “syngameon” together with a few other western European white oaks (Cannon and Petit, 2019). A syngameon is a complex of species exchanging genes, while still maintaining some degree of interspecific diversity enabling to differentiate, on the basis of leaf morphology for instance, the originally described species (Kremer et al, 2002 among many others dealing with the identification of oak species on the basis of their phenotype). The full sequencing of the genome of Q. robur (Plomion et al, 2018) opened an avenue for the development of new genomic tools to investigate the concept of species in this syngameon and to identify gene flows between populations and species. To date, whole genome sequencing and annotation is completed for Q. robur, Q. lobata Née, Q. suber L., and Fagus sylvatica L. (Plomion and Martin, 2020).
The preprint of Lang et al (2021) builds up on this background and provides a very interesting and important contribution to this area. First, it provides access to a very large data base of genomic sequences, based on 800 gene fragments and identified 12500 Single Nucleotide Polymorphisms (SNPs) as well as 1500 insertions deletions. 11 individuals assigned to the species Q. robur, 13 assigned to Q. petraea and 1 to Q. ilex L. were used to establish this data base. This is an important asset for developing future research on species identification based on their genomes. In full agreement to the policy of all PCIs about data availability, full access is provided to the data under a free use license CC-BY.
The manuscript has already a long history as a preprint and it was not that easy to find reviewers willing to do the job of reviewing such a large amount of information with a lot of technical details. At the end, we succeeded and obtained three very enthusiastic and positive reviews (attached to this recommendation). All reviewers agreed that this was an important contribution in the field, insisted on the quality of the writing and of the provided data, and finally agreed that the manuscript was to be recommended after a careful revision. The revision was readily done by the authors. One of the reviewers found that the preprint is close to a “data paper”, i.e., a paper providing the required information to enable (and advertise) the reuse of an important data set, which is only partly true. Indeed, the authors nicely used their data to address one of the most debated questions about the population genetics: how comes that in this syngameon, with inter-fertile individuals, clearly distinguishable species (from the phenotype at least) have been maintained?
The main result of the preprint is that indeed there are some (only few) genomic differences among the two species in the patterns of distribution of the SNPs, but also that the patterns reveal a clear introgression of the genome of one of the species into the other one. This was based on a Bayesian clustering analysis reported figure 3, evidencing that 2 individuals displaying an unambiguous Q. petraea phenotype could not be assigned to any of the species. The usual hypothesis postulates an introgression of the Q. robur genome, the pioneer species able to colonize open areas during the post-glacial recolonization into that of Q. petraea, due to hybridization and successive events of back gross with inflowing Q. petraea pollen. Such an introgression was hypothesized decades ago, and receives here some degree of confirmation. Recent findings showed the importance of such an introgression for the adaptation of local oak populations to altitudinal gradients (Leroy et al, 2020).
Given the importance of the contribution (both from the point of view of the development of new genomic tools and from that of our understanding of the population genetics in the oak syngameon, I do recommend this preprint in the frame of the PCI Forest and Wood Sciences after an insightful peer reviewing and a very careful revision leading to the current version 4 of this preprint.
References
Cannon CH, Petit RJ (2020) The oak syngameon: more than the sum of its parts. New Phytologist, 226, 978–983. https://doi.org/10.1111/nph.16091
Kremer A, Dupouey JL, Deans JD, Cottrell J, Csaikl U, Finkeldey R, Espinel S, Jensen J, Kleinschmit J, Dam BV, Ducousso A, Forrest I, Heredia UL de, Lowe AJ, Tutkova M, Munro RC, Steinhoff S, Badeau V (2002) Leaf morphological differentiation between Quercus robur and Quercus petraea is stable across western European mixed oak stands. Annals of Forest Science, 59, 777–787. https://doi.org/10.1051/forest:2002065
Lang T, Abadie P, Léger V, Decourcelle T, Frigerio J-M, Burban C, Bodénès C, Guichoux E, Provost GL, Robin C, Tani N, Léger P, Lepoittevin C, Mujtar VAE, Hubert F, Tibbits J, Paiva J, Franc A, Raspail F, Mariette S, Reviron M-P, Plomion C, Kremer A, Desprez-Loustau M-L, Garnier-Géré P (2021) High-quality SNPs from genic regions highlight introgression patterns among European white oaks (Quercus petraea and Q. robur). bioRxiv, 388447, version 4 peer-reviewed and recommended by PCI Forest and Wood Sciences. https://doi.org/10.1101/388447
Leroy T, Louvet J-M, Lalanne C, Provost GL, Labadie K, Aury J-M, Delzon S, Plomion C, Kremer A (2020) Adaptive introgression as a driver of local adaptation to climate in European white oaks. New Phytologist, 226, 1171–1182. https://doi.org/10.1111/nph.16095
Plomion C, Aury J-M, Amselem J, Leroy T, Murat F, Duplessis S, Faye S, Francillonne N, Labadie K, Le Provost G, Lesur I, Bartholomé J, Faivre-Rampant P, Kohler A, Leplé J-C, Chantret N, Chen J, Diévart A, Alaeitabar T, Barbe V, Belser C, Bergès H, Bodénès C, Bogeat-Triboulot M-B, Bouffaud M-L, Brachi B, Chancerel E, Cohen D, Couloux A, Da Silva C, Dossat C, Ehrenmann F, Gaspin C, Grima-Pettenati J, Guichoux E, Hecker A, Herrmann S, Hugueney P, Hummel I, Klopp C, Lalanne C, Lascoux M, Lasserre E, Lemainque A, Desprez-Loustau M-L, Luyten I, Madoui M-A, Mangenot S, Marchal C, Maumus F, Mercier J, Michotey C, Panaud O, Picault N, Rouhier N, Rué O, Rustenholz C, Salin F, Soler M, Tarkka M, Velt A, Zanne AE, Martin F, Wincker P, Quesneville H, Kremer A, Salse J (2018) Oak genome reveals facets of long lifespan. Nature Plants, 4, 440–452. https://doi.org/10.1038/s41477-018-0172-3
Plomion C, Martin F (2020) Oak genomics is proving its worth. New Phytologist, 226, 943–946. https://doi.org/10.1111/nph.16560
A new mechanism for tree mortality due to drought and heatwaves
A very useful simulation of the impact of drought and enhanced temperatures on embolism in trees and on tree dieback
Recommended by Erwin Dreyer based on reviews by Sabine Rosner and 1 anonymous reviewerWater availability has been known to strongly modulate forest productivity and tree growth on an interannual basis (as revealed by numerous dendrochronological studies) and across biomes (Ellison et al, 2017). Recurrent episodes of severe drought lead to decreased soil water content and as a consequence to visible losses in annual growth increment, and in some cases even to tree death and forest decline. The occurrence of such drought events and of larger scale tree dieback, seem to be increasing over the last decades, albeit such processes are not new. The causes for drought-induced tree death are still disputed; in many cases, tree death occurs after the release of drought, and is caused by severe attacks by pests and pathogens. In other cases, tree death is caused by recurrent drought events over several years, leading to a depletion of stored carbohydrates, growth decline and ultimately death.
However, this understanding of drought-induced tree dieback, which applies to drought events that occurred in temperate climate biomes during the end of the 20th century, seems inadequate to explain the increasing occurrence of large scale dieback induces by recent drought episodes (Allen et al, 2015). In these recent cases a direct impairment of hydraulic functions seems responsible for tree death. Such impairments (cavitation and resulting massive embolism) have been well documented through extensive research that started in the 90s. Up to now, the consensus was that trees are fairly well protected against such potentially lethal dysfunctions: an efficient stomatal closure limits transpiration and the risk of runaway embolism. Many tree models based on the known hydraulic properties of trees (vulnerability of different organs to cavitation, hydraulic conductance of these organs, transpiration, stomatal conductance…) were developed since the seminal work of Tyree and Sperry (1989) and only seldom predicted the occurrence of runaway embolism (Cochard and Delzon, 2013).
These models considered the impact of drought through reduced soil water availability, which is indeed the central process during drought, but overlooked to some extent the fact that drought is frequently and increasingly associated to higher temperatures, which may change rather severely model parameters and result in a higher risk of runaway embolism.
The present preprint proposed by Cochard (2020) bases on such a new hydraulic model (the model SurEau, Martin StPaul et al, 2017; Cochard et al, 2020) integrating more explicitly the impact of temperature on different parameters. Two parameters appear particularly relevant and highly sensitive to temperature:
(i) the vapor pressure deficit of the air (VPD), which increases exponentially with temperature and results in increased transpiration and more rapid soil water depletion; this effect is well known and has been the matter of many research and modelling;
(ii) the cuticular conductance to water vapor, which becomes the most important limit to transpiration once stomata are closed, and which is much less well documented with respect to mean values and temperature sensitivity (mainly because this process is difficult to record). Recent advances (Schuster et al, 2016) provided some insight into the importance of this parameter and showed how it may rapidly increase with temperature (see references in the preprint).
The presented work bases on this new model to document more precisely how enhanced temperature may increase water loss through transpiration and consequently induce runaway embolism in trees more rapidly than usually expected. The hypothesis that the temperature response of cuticular conductance may play a central role in the sensitivity of trees to a combination of soil water depletion and enhanced air (and leaf) temperature was tested through numerical simulations with SurEau. The results are very clear: temperature-dependent increases in cuticular conductance may accelerate the onset of runaway embolism at a rate that was not expected before.
The demonstration is indeed very clear and convincing. It remains however a simulation (or an “in silico experiment”. Data providing real values of cuticular conductance remain scarce, and data documenting its response to enhanced temperatures even scarcer. This opens an avenue for new research and investigations, and Cochard (2020) provides some clues about which data and which experiments could confirm the central role of temperature induced changes in cuticular conductance with temperature (eg new measurements of Tp, the phase transition temperature that matches the range of temperatures known to trigger mortality during hot-droughts, Billon et al. (2020)).
I believe this preprint is an important contribution in this field, and the reviewers were of the same opinion (see their reviews attached to this recommendation). Indeed, this preprint illustrates how simulation exercises can help us identify some key processes that require further attention and documentation. I believe this is an important contribution to our understanding of the rapid, drought-induced tree death observed in different parts of the world at alarming rates.
As such, and combined with a detailed description of the model SurEau, this preprint is a very important addendum to the corpus of knowledge that is currently gathered around the hydraulic functioning of trees.
References
Allen, C. D., Breshears, D. D., and McDowell, N. G. (2015). On underestimation of global vulnerability to tree mortality and forest die‐off from hotter drought in the Anthropocene. Ecosphere, 6(8), 1-55. doi: https://doi.org/10.1890/ES15-00203.1
Billon et al. (2020). The DroughtBox: A new tool for phenotyping residual branch conductance and its temperature dependence during drought. Plant, Cell and Environment, 43, 1584-1594. doi https://doi.org/10.1111/pce.13750
Cochard, H. (2020) A new mechanism for tree mortality due to drought and heatwaves. bioRxiv, 531632, ver. 2 peer-reviewed and recommended by PCI Forest and Wood Sciences. doi: https://doi.org/10.1101/531632
Cochard, H., Martin-StPaul, N., Pimont, F., and Ruffault, J. (2020). SurEau.c: a mechanistic model of plant water relations under extreme drought. bioRxiv, 2020.05.10.086678. doi: https://doi.org/10.1101/2020.05.10.086678
Ellison et al. (2017). Trees, forests and water: Cool insights for a hot world. Global Environmental Change, 43, 51-61. doi: https://doi.org/10.1016/j.gloenvcha.2017.01.002
Martin‐StPaul, N., Delzon, S., and Cochard, H. (2017). Plant resistance to drought depends on timely stomatal closure. Ecology letters, 20(11), 1437-1447. doi: https://doi.org/10.1111/ele.12851
Schuster et al. (2016). Effectiveness of cuticular transpiration barriers in a desert plant at controlling water loss at high temperatures. AoB Plants, 8(1), plw027. doi: https://doi.org/10.1093/aobpla/plw027
Tyree, M. T., and Sperry, J. S. (1989). Vulnerability of xylem to cavitation and embolism. Annual review of plant biology, 40(1), 19-36. doi: https://doi.org/10.1146/annurev.pp.40.060189.000315